Engineering the Stem Cell Niche
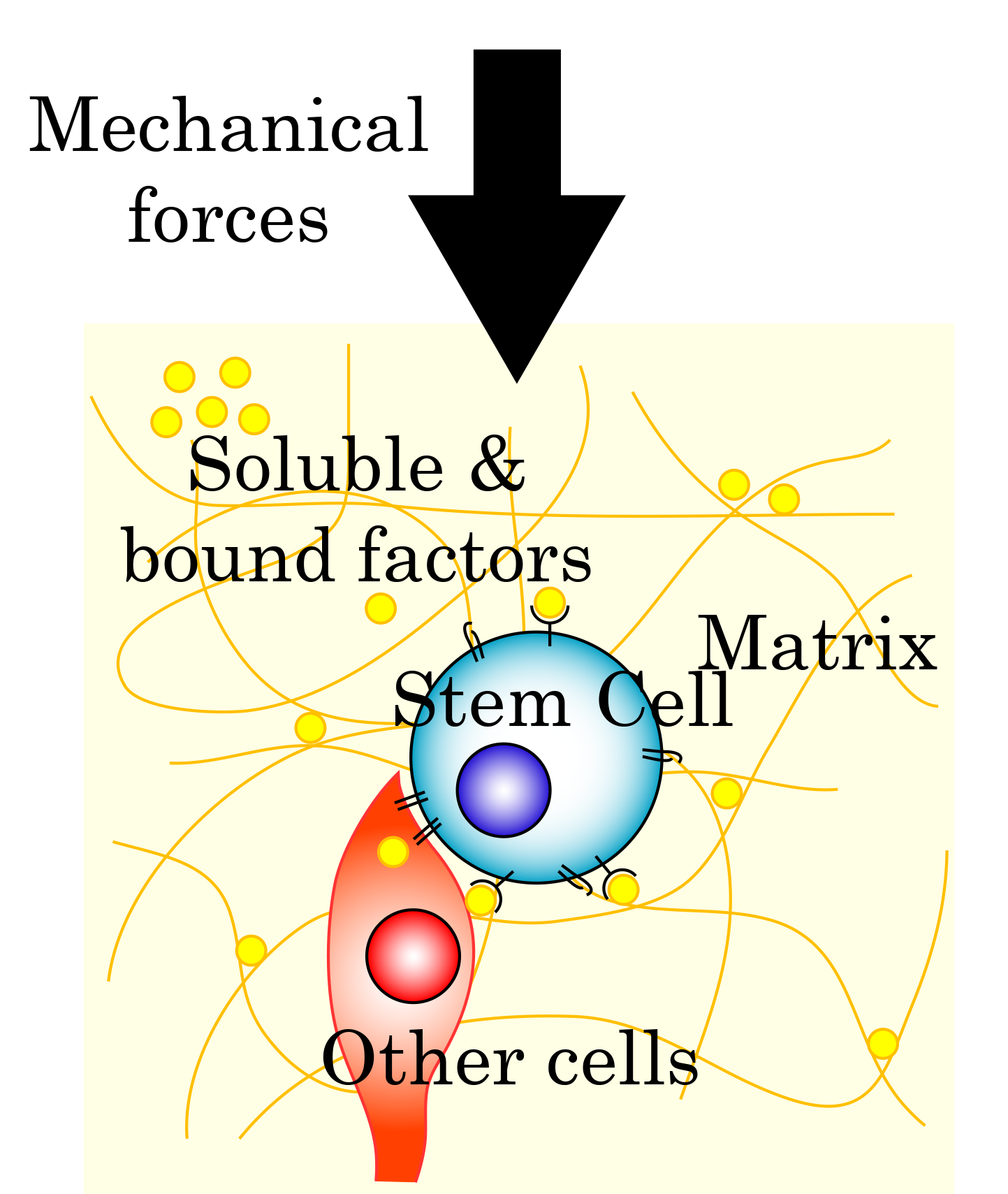
A unifying theme across all of our research is the engineering of microenvironmental cues that actively shape cell function. We are fundamentally interested in how the physical, chemical, and biological properties of a cell's immediate environment — its niche — govern proliferation, adhesion, migration, differentiation, and phenotypic maintenance. To study and exploit these relationships, our group has developed a versatile family of hydrogel biomaterials that can be precisely tuned in stiffness, porosity, oxygen delivery capacity, and bioactive signaling content.
A signature capability of our laboratory is the the incoporation and biocongugation of bioactive molecules — growth factors, cytokines, axon guidance proteins, and fluorocarbons — directly within these 3D hydrogel polymer networks. This approach moves beyond passive scaffolds to create materials that actively instruct cells how to behave, providing sustained, spatially defined signals that soluble delivery cannot replicate. We apply these engineered niches across three interconnected research programs: liver organoid platforms for metabolic disease modeling, spinal cord injury repair and regeneration, and oxygenating/antibacterial hydrogels for chronic wound healing.
Organoids & 3D Disease Modeling
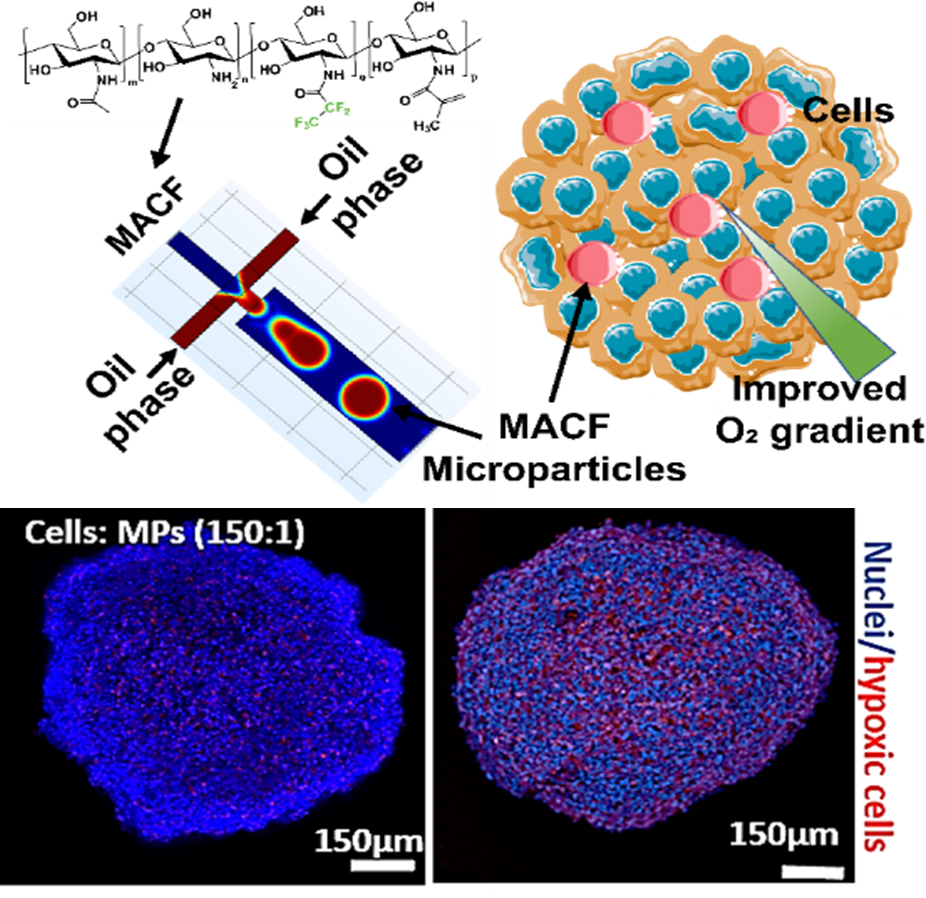
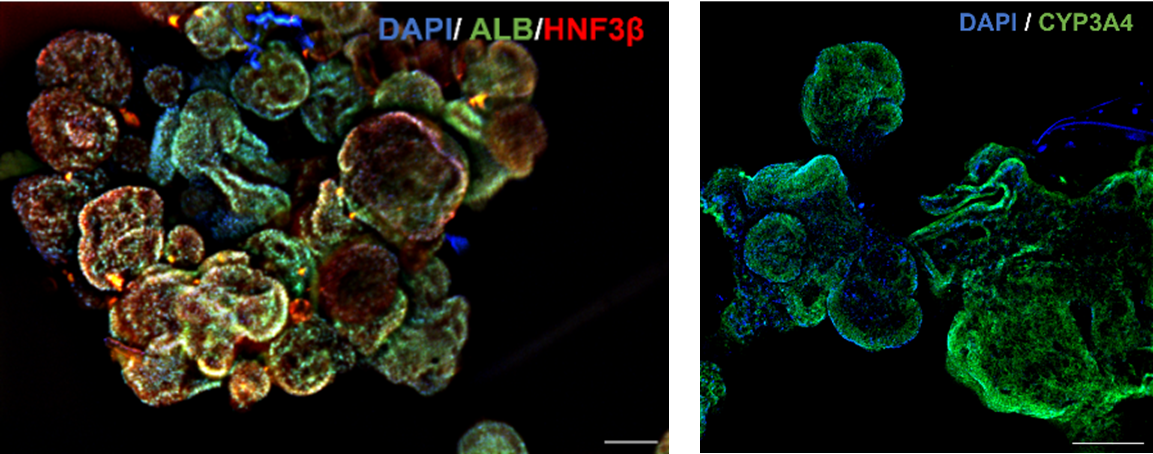
We develop oxygenating biomaterial platforms based on methacrylamide chitosan modified with perfluorocarbons (MACF) to create physiologically relevant 3D liver models for drug toxicity screening and disease modeling. Perfluorocarbon-loaded microparticles functionalized with liver ECM proteins dramatically improve hepatocyte survival, albumin secretion, and CYP450 activity in large spheroid cultures that would otherwise become hypoxic.
In collaboration with the Cleveland Clinic, we demonstrated that local oxygen tension is a primary determinant of hepatotoxin detection sensitivity — shifting design principles for in vitro liver systems. Current work has shifted to using pluripotent stem cells to gernate liver organoids to target mechano-oxygen crosstalk in liver development and disease progression, building time-evolving hydrogel platforms with independently tunable mechanics and oxygen delivery to dissect mechanisms of metabolic liver disease at the molecular level.
We also develop cerebral 'brain' organoid models using pluripotent stem cells — specifically choroid plexus organoids — as physiologically relevant platforms to study CNS barrier functions. These self-organizing 3D structures recapitulate choroid plexus epithelial architecture and barrier functions, providing a human-tissue-mimetic system for investigating CNS barrier integrity, transport, and diseases.
Selected Publications
- Mansouri M, Imes WD, Roberts OS, Leipzig ND. Fabrication of oxygen carrying microparticles functionalized with liver ECM-proteins to improve phenotypic 3D in vitro liver assembly, function, and responses. Biotechnology and Bioengineering. 2023. doi: 10.1002/bit.28456.
- Mansouri M, Lamichhane A, Das D, Aucejo F, Tavana H, Leipzig ND. Comparison of engineered liver 3D models and the role of oxygenation for patient-derived tumor cells and immortalized cell lines in the detection of hepatotoxins. Advanced Biology. 2024;8(2):e2300386.
- Mansouri M, Beemer S, Kothapalli CR, et al. Generation of fluorinated oxygenating methacrylamide chitosan microparticles to increase cell survival and function in large liver spheroids. ACS Applied Materials & Interfaces. 2022;14(4):4899–4913.
- Qureshi AA, et al., Leipzig N, et al. Tumor organoids for primary liver cancers: A systematic review. JHEP Reports. 2024;6(12):101164.
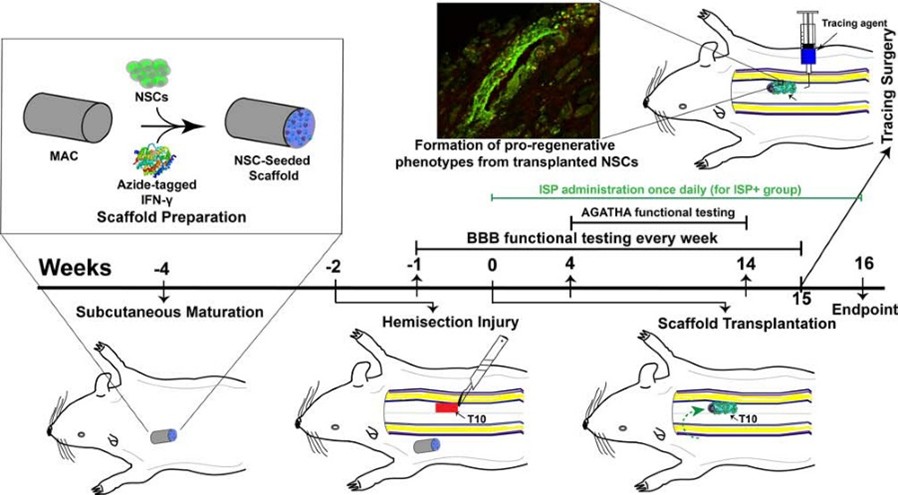
CNS Injury, Repair & Regeneration
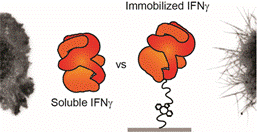
Spinal cord injury (SCI) demands multifactorial repair strategies that simultaneously address inflammation, glial scarring, and loss of structural continuity. Our laboratory designs biomimetic hydrogel scaffolds with covalently tethered growth factors and cytokines that provide spatially precise, sustained signaling to cells in ways that soluble delivery cannot replicate.
We have demonstrated that subcutaneous pre-conditioning of growth factor-functionalized scaffolds before spinal implantation leverages the host foreign body response to mature the cellular microenvironment and significantly improve functional recovery. Current DoD SCIRP-funded work pursues a combinatorial therapy combining optimized human ESC-derived neural stem cell grafts subcutaneously preconditioned in our engineered scaffolds with NVG-291, a peptide that promotes neurogenesis and neural circuit repair. A parallel NSF-funded collaboration with Cleveland State University uses quantitative mechanobiological characterization of the chronic SCI scar to rationally design mechanically matched implants and formulate novel strategies to address glial and fibrotic processes. Additional research in the lab studies post-traumatic syringomyelia, where we identified dysregulated betaine osmoregulation as a novel therapeutic target.
Selected Publications
- Bradshaw KJ, Leipzig ND. Applications of regenerative tissue-engineered scaffolds for treatment of spinal cord injury. Tissue Engineering Part A. 2025. doi: 10.1089/ten.tea.2024.0194.
- Pukale DD, Adkins-Travis K, Aryal SR, et al. Investigating post-traumatic syringomyelia and local fluid osmoregulation via a rat model. Fluids and Barriers of the CNS. 2024;21(1):19.
- Baumann HJ, Mahajan G, Ham TR, et al. Softening of the chronic hemisection SCI scar parallels dysregulation of cellular and ECM content. J Mech Behav Biomed Mater. 2020;110:103953.
- Ham TR, Pukale DD, Hamrangsekachaee M, Leipzig ND. Subcutaneous priming of protein-functionalized chitosan scaffolds improves function following SCI. Mater Sci Eng C. 2020;110:110656.
- Ham TR, Farrag M, Leipzig ND. Covalent growth factor tethering to direct neural stem cell differentiation and self-organization. Acta Biomaterialia. 2017;53:140–151.
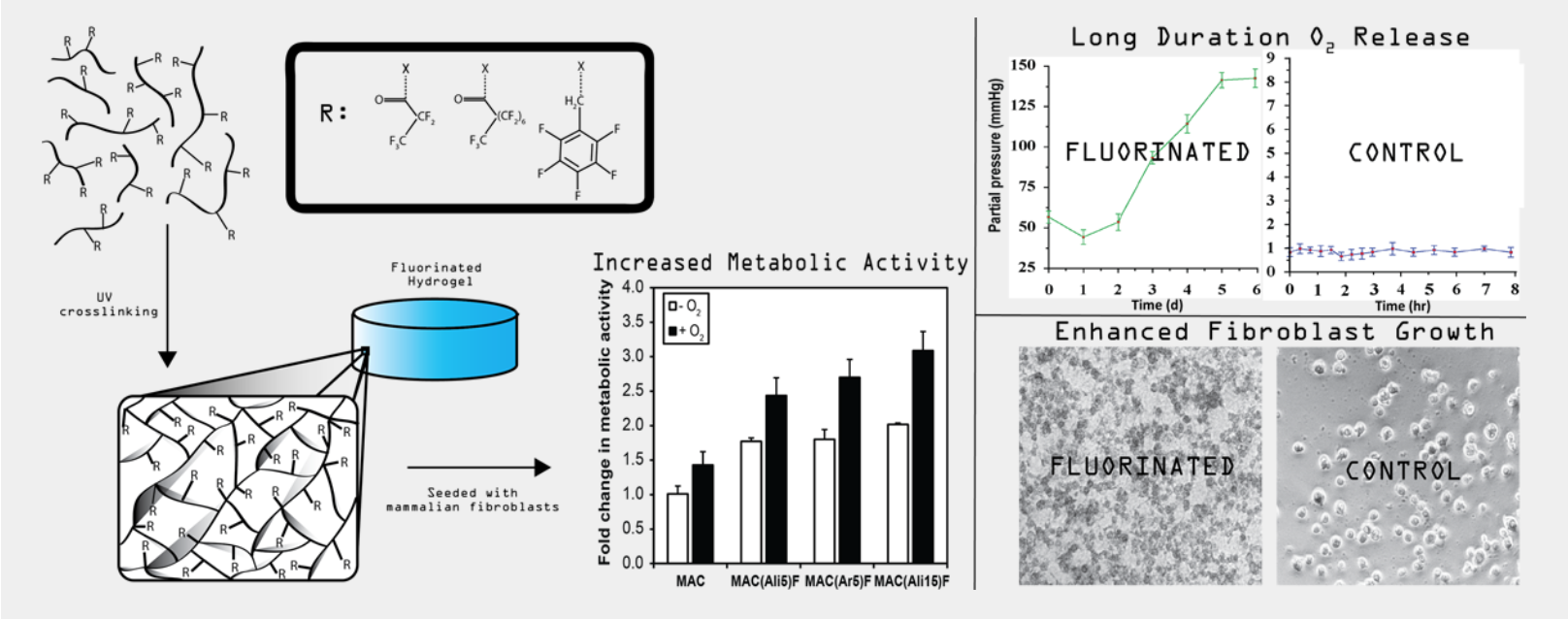
Wound Healing & Infection Control
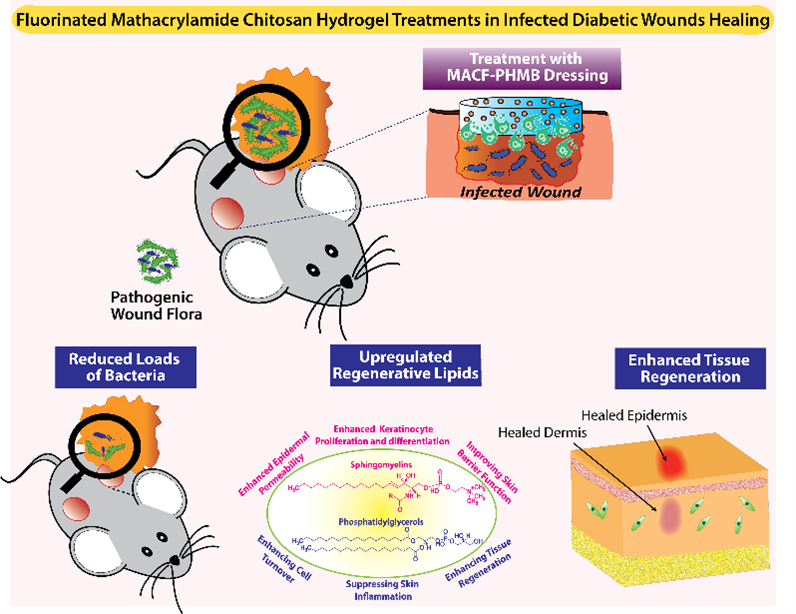
Chronic diabetic wounds are characterized by severely depleted tissue oxygen (pO2 as low as 5 mmHg), which impairs metabolism, collagen synthesis, angiogenesis, and antimicrobial defense. We invented MACF hydrogel dressings that covalently incorporate perfluorocarbons to sustain local oxygen delivery for up to five days, improving collagen organization, re-epithelialization, and ECM quality in both porcine and murine wound models without an external oxygen source.
Building on this foundation, we developed next-generation multifunctional dressings incorporating heparin-chitosan polyionic complex nanoparticles loaded with broad-spectrum antibiotics, simultaneously oxygenating the wound bed and combating bacterial biofilm to break the infection-hypoxia cycle that perpetuates chronic wounds. Most recently, we demonstrated that MACF treatment fundamentally restores the structural quality of regenerated ECM in chronically infected wounds. Ongoing work is advancing bacteriophage-delivering MACF hydrogels as a next-generation strategy against antibiotic-resistant pathogens.
Selected Publications
- Durr HA, Abri S, Salinas SD, et al. Extracellular matrix repair and organization of chronic infected diabetic wounds treated with MACF hydrogels. Acta Biomaterialia. 2025;199(1):166–177.
- Abri S, Durr H, Barton HA, et al. Chitosan-based multifunctional oxygenating antibiotic hydrogel dressings for managing chronic infection in diabetic wounds. Biomaterials Science. 2024;12(13):3458–3470.
- Abri S, Attia R, Pukale DD, Leipzig ND. Modulatory contribution of oxygenating hydrogels and PHMB on the antimicrobial potency of neutrophil-like cells. ACS Biomater Sci Eng. 2022;8(9):3842–3855.
- Durr H, Leipzig ND. Advancements in bacteriophage therapies and delivery for bacterial infection. RSC Materials Advances. 2023;4:1249–1257.
- Patil PS, Fountas-Davis N, et al. Fluorinated methacrylamide chitosan hydrogels enhance collagen synthesis in wound healing through increased oxygen availability. Acta Biomaterialia. 2016;36:164–174.
